 |

The study of the surface composition of a material is not always sufficient. In the case of ion implantation or of thin film or passivation layer deposits, it becomes necessary to know the chemical composition of the inner layers. To reach these layers, we use an etching ion gun to clean the specimen surface more or less quickly. This technique is a complement to XPS and AES and allows to profiling the concentration of the species making up the material. The next section describes the means used in the ionic implantation example.

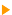 Ion etching Principle. Ion etching Principle.
The ion beam is obtained by collision between a gas and electrons. These ions are accelerated and bombard the surface of the specimen. The ion impacts on the material pull out the superficial atoms. This etching reveals a new layer which is then characterized by XPS or AES spectroscopy.
Some instruments are equipped with a visual display system enabling to obtain an ion image. This allows a better definition of the etched area and allows the best possible fit to the studied area.
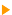 Ion gun adjustment. Ion gun adjustment.
The efficiency of a gun depends on several parameters:
To adjust an ion gun, we measure the current collected from the specimen. Needle valve is used to introduce the gas into the gun. This allows a precise pressure adjustment. By increasing the gas pressure, the electron/gaz interaction is enhanced and the ion quantity grows. When this pressure becomes too high, a rearrangement phenomenon occurs; the ions are neutralized. Regardless of your system, the gun pressure should not exceed a few mbar. In the vacuum system, beyond 10-7 mbar, damages to the various elements building up the instrument do occur. Electrons are obtained from a filament by thermoelectronic emission. The principle is the following: A current flows through a filament and an outside potential extracts some of the emited electrons. The more the emission current of the filament increases and the more ions are obtained. This phenomenon has a limit since beyond a certain intensity, a current increase does not generate more electrons, there is saturation of the emission. When the saturation point is exceeded, the filament lifetime becomes very short. The electron/gaz interaction generates the ions. These ions are accelerated and focused into a beam which can then be swept. At constant ion flow, the swept surface is inversely proportional to the current density. We can conclude that the etching speed will decrease as the etched surface becomes larger. The gun is fixed versus the instrument and the variation of the ion incidence angle is accomplished by tilting the specimen. When the gun is perpendicular to the specimen, the etching speed is high and the surface roughness increases. On the contrary, at a small incidence angle, the etching speed and roughness are decreasing. 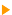 Ion implantation: XPS concentration profile. Ion implantation: XPS concentration profile.
The ion bombardment of the surface of a substance induces some modification of this substance surface chemical and physical properties. In case of important incident energy, the ions penetrate in the matter and stay inserted between the atoms: this is referred to as ion implantation. These more or less important changes can be studied by surface spectroscopy coupled with ion bombardment.
The following animation summarizes this characterization protocol: XPS / Ion etching.  Analysis protocol We polished a steel pellet and implanted molybdenum. The specimen is introduced in the analysis chamber and is cooled down with liquid nitrogen. Etching is carried out with a current density of 2 a/cm². 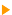 Results Results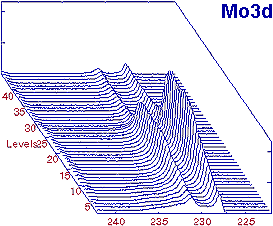 Profile of the Mo3d level We established the chemical composition of each level and starting from these results, we plotted the concentration profiles of each species. These profiles show an enrichment in molybdenum induced by the preferential etching of the other elements. The studies carried out with characterization angles of 60° and 80° showed that this enrichment was located at the very surface with a depth of a few angstroms.  Conclusions. Conclusions.Ion etching enables to study the inner layers of a specimen. The etching speed of the elements differs according to the specimen composition. We call this preferential etching. |
|||||||||||||